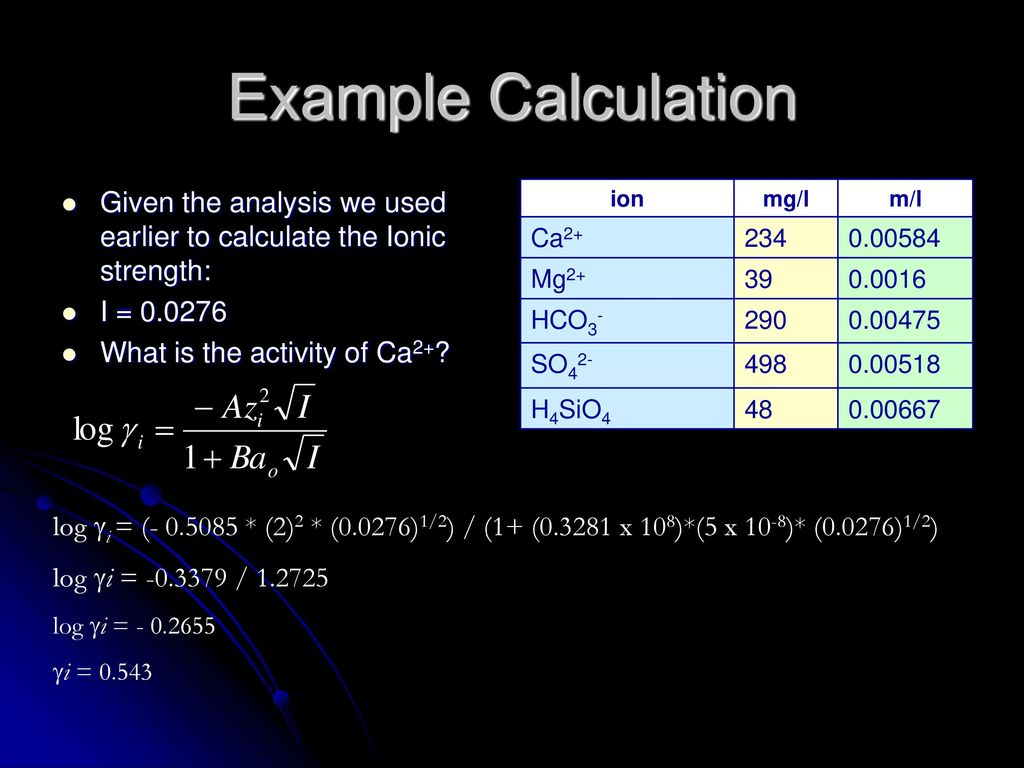
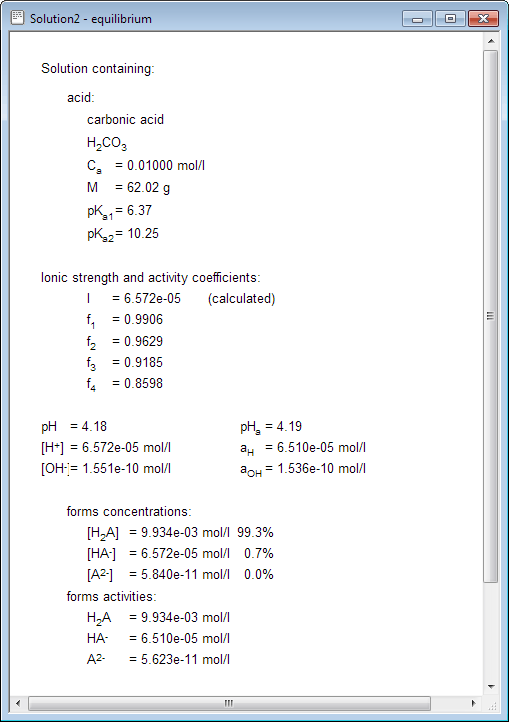
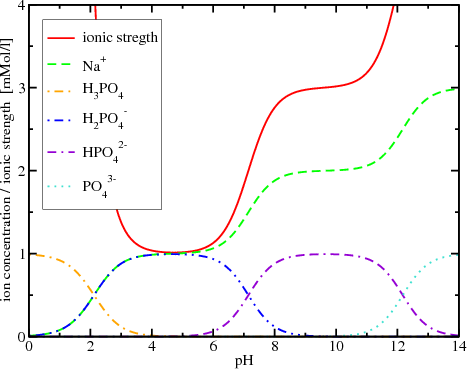
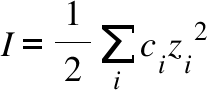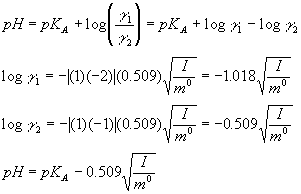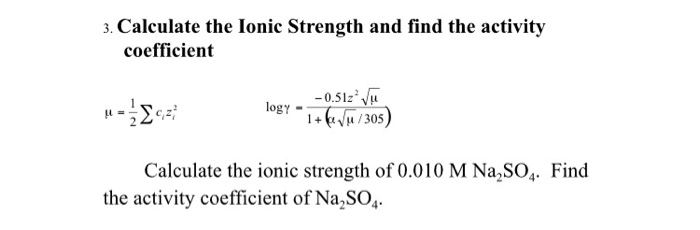CHAPTER 5 ELECTROLYTE EFFECTS AND EQUILIBRIUM: CALCULATIONS IN COMPLEX SYSTEMS Introduction to Analytical Chemistry ppt download
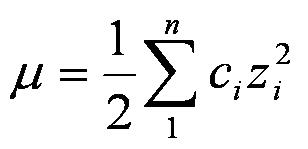
pH calculations and more in fundamentals of pharmaceutics. : What is ionic strength of solutions and how is it calculated?

SOLVED: Calculate or describe the indicated quantity: a) The ionic strength of a solution that is 0.1 m in MgCl2 b) the mean activity a+ of magnesium chloride in terms of its